
Steam Pipes - Online Pressure drop CalculatorĬalculate pressure drops in steam distribution pipe lines. Steam systems carries heat through pipes from the boiler to consumers as heat exchangers, process equipment etc. The amount of flash steam generated depends on steam pressure and pressure in the condensate lines. Vapor and steam enthalpy, specific enthalpy of saturated liquid, saturated vapor and superheated vapor.īasic steam thermodynamics - entropy diagram. Steam table with sensible, latent and total heat, and specific volume at different gauge pressures and temperatures. Saturated Steam - Properties - Imperial Units Saturated Steam Table with steam properties as specific volume, density, specific enthalpy and specific entropy. 
Vapor and saturation pressure for some common liquids. Latent heat is the heat when supplied to or removed from air results in a change in moisture content - the temperature of the air is not changed. Tutorial to the basic physics behind flash steam generation Thermodynamics of steam and condensate systems.Ĭalculate flash steam generation in condensate pipe lines.Ĭontrol valves and cavitation, application ratio and multi stage control valves.įlash Steam Generation - Fundamental Physics Steam & condensate systems- properties, capacities, pipe sizing, systems configuration and more. With higher pressure - more energy can be transferred in a steam distribution system. Note! The specific volume of steam decreases with increased pressure - and the amount of heat energy distributed by the same volume increase.The evaporation heat is 2046.53 kJ/kg at 7 bar g. The specific enthalpy of vaporization decreases with steam pressure. The heat energy (enthalpy of evaporation) needed at 7 bar g to vaporize the water to steam is actually less than required at atmospheric pressure. According the table 720.94 kJ is required to raise 1 kg of water from 0 oC to saturation temperature 170 oC. More heat energy is required to raise its temperature to saturation point at 7 bar g than needed when the water is at atmospheric pressure. In a steam distribution system the pressure is always more than 0 bar gauge.Īt 7 bar g ( absolute 8 bar) the saturation temperature of water is 170.42 oC. Steam at atmospheric pressure is of a limited practical use because it cannot be conveyed by its own pressure along a steam pipe to the points of use. = 2675.43 kJ/kg Example - Boiling Water at 170 oC and 7 bar (700 kPa) Atmospheric Pressure 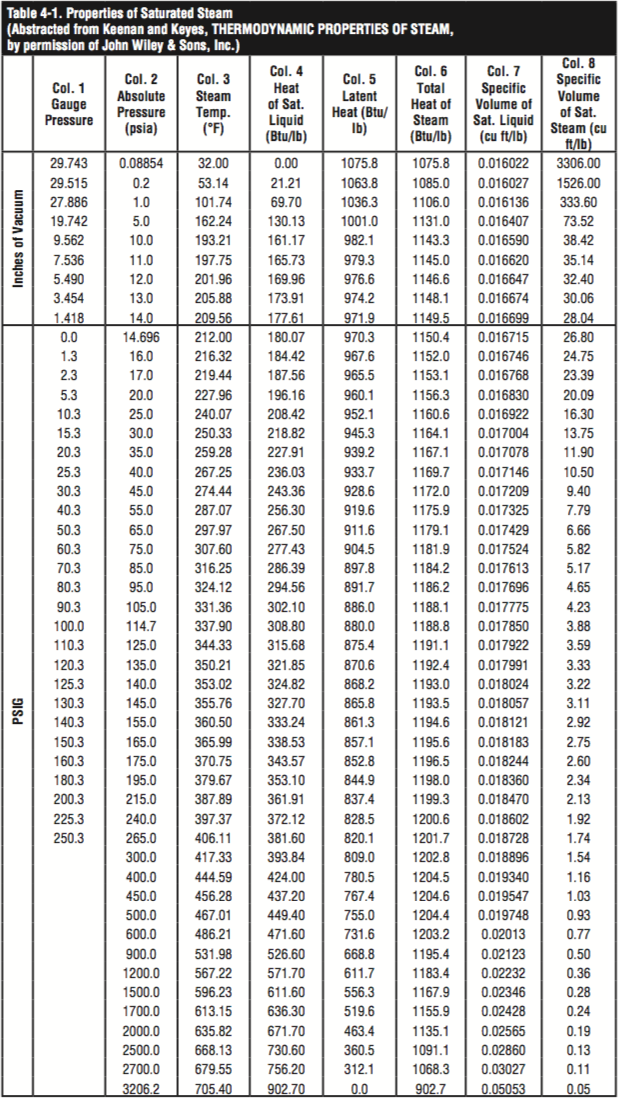
The total specific enthalpy for steam at 0 bar gauge is: Therefore at 0 bar g ( absolute 1 bar) the specific enthalpy of vaporation is 2257.19 kJ/kg. Therefore the specific enthalpy of water at 0 bar g (absolute 1 bar ) and 100 oC is 417.51 kJ/kg.Īnother 2257.92 kJ of energy is required to evaporate 1 kg of water at 100 oC into 1 kg of steam at 100 oC.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
